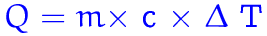Thermochemistry
Thermodynamics is the study of energy and its transformations. When studying thermochem, a major focus is heat and its behavior. There are two forms of energy: Kinetic energy, the
energy of motion, and potential energy, the energy that an object posses by its position
relative to other objects.
SI units for energy
Joule(J)
Calorie(cal): 1 cal=4.184J
*different from a nutritional Calorie(Cal).
Thermodynamic Properties- A system is a specific amount of matter and the surroundings are everything outside the system. During a reaction energy is exchanged between the
surroundings and the system.
Work is a result of a force moving an object a certain distance.
Work can be found using this formula: force times distance.
Heat is energy that is transformed as a consequence of
temperature differences.
Energy is the capacity to do work and to transfer heat.
The First law of Thermodynamics states that the change in internal energy of a system, Delta E, is the sum of the heat, q, transfered in to our system and the work done on or
by the system. In other words, heat is a form of energy and can neither be created
or destroyed. When a system absorbs heat it is known as an endothermic process.
When a system releases heat it is known as an exothermic process.
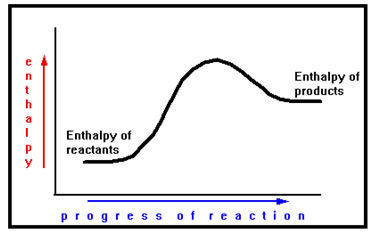
In a chemical process, the enthalpy of a reaction is determined by the following equation:
enthalpy of reaction =enthalpy of products- enthalpy of reactants
Thermodynamics is the study of energy and its transformations. When studying thermochem, a major focus is heat and its behavior. There are two forms of energy: Kinetic energy, the
energy of motion, and potential energy, the energy that an object posses by its position
relative to other objects.
SI units for energy
Joule(J)
Calorie(cal): 1 cal=4.184J
*different from a nutritional Calorie(Cal).
Thermodynamic Properties- A system is a specific amount of matter and the surroundings are everything outside the system. During a reaction energy is exchanged between the
surroundings and the system.
Work is a result of a force moving an object a certain distance.
Work can be found using this formula: force times distance.
Heat is energy that is transformed as a consequence of
temperature differences.
Energy is the capacity to do work and to transfer heat.
The First law of Thermodynamics states that the change in internal energy of a system, Delta E, is the sum of the heat, q, transfered in to our system and the work done on or
by the system. In other words, heat is a form of energy and can neither be created
or destroyed. When a system absorbs heat it is known as an endothermic process.
When a system releases heat it is known as an exothermic process.
In a chemical process, the enthalpy of a reaction is determined by the following equation:
enthalpy of reaction =enthalpy of products- enthalpy of reactants
The enthalpies of reaction follow some simple rules:
1. The enthalpy of reaction is proportional with the amount of reactant that is reacting.
2. If you reverse a reaction the sign of enthalpy will change.
3. The enthalpy of reaction depends on the physical state that the products and reactants are in.
Heat Capacity
Heat Capacity is the amount of heat that is required to raise its temperature by 1 K.
For one mole of pure substance the heat capacity is known as its molar heat capacity: the energy
required to raise the temperature of one mole of a substance by one degree Celsius.
q=mc(change in temperature)
q= the amount of heat
c= the specific heat
(the amount of heat absorbed by a substance)
m= mass
change in heat= temperature final- temperature initial.
1. The enthalpy of reaction is proportional with the amount of reactant that is reacting.
2. If you reverse a reaction the sign of enthalpy will change.
3. The enthalpy of reaction depends on the physical state that the products and reactants are in.
Heat Capacity
Heat Capacity is the amount of heat that is required to raise its temperature by 1 K.
For one mole of pure substance the heat capacity is known as its molar heat capacity: the energy
required to raise the temperature of one mole of a substance by one degree Celsius.
q=mc(change in temperature)
q= the amount of heat
c= the specific heat
(the amount of heat absorbed by a substance)
m= mass
change in heat= temperature final- temperature initial.
Hess's law
Hess's Law states that if a series of reactions are added together, the enthalpy change for the total reaction is the sum of the enthalpy changes for the individual steps.
The change in heat will be equal to the sum of enthalpy changes for the steps.
Hess's Law states that if a series of reactions are added together, the enthalpy change for the total reaction is the sum of the enthalpy changes for the individual steps.
The change in heat will be equal to the sum of enthalpy changes for the steps.
Picture citations:
Picture 1: Picture 2: N.d. Tutorvista. Web. 29 May 14. <http://images.tutorvista.com/cms/images/83/formula-for-heat-transfer.png>.
N.d. Web. 29 May 14. <http://dl.clackamas.edu/ch105/lesson%208%20images/enth09.jpg>.
Picture 3: N.d. Purdue Education. Web. <http://www.chem.purdue.edu/gchelp/howtosolveit/Thermodynamics/ThermoArt/Hess_law_pure.JPG>.
Picture 1: Picture 2: N.d. Tutorvista. Web. 29 May 14. <http://images.tutorvista.com/cms/images/83/formula-for-heat-transfer.png>.
N.d. Web. 29 May 14. <http://dl.clackamas.edu/ch105/lesson%208%20images/enth09.jpg>.
Picture 3: N.d. Purdue Education. Web. <http://www.chem.purdue.edu/gchelp/howtosolveit/Thermodynamics/ThermoArt/Hess_law_pure.JPG>.